Click here to see all images
September, 2023
Case of the Month
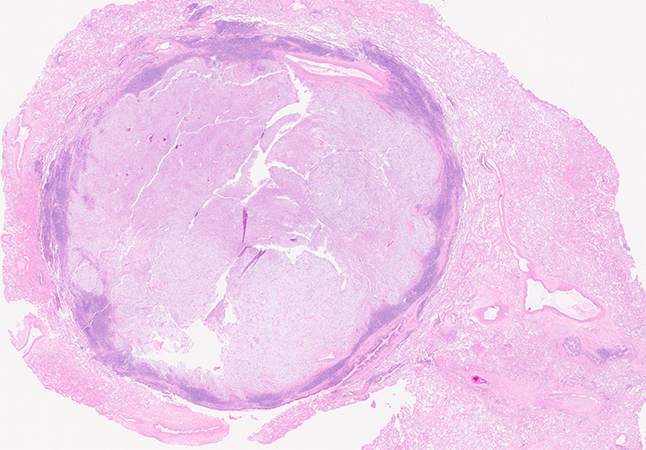
Clinical History: A middle-aged woman with never smoking history complained of chest pain and underwent a chest CT scan. She has no relevant past medical or familial history and no significant environmental exposure. An incidental, solitary 1.5 cm round nodule was found in the periphery of the left upper lobe of lung. A left upper lobe wedge resection was performed for diagnostic and treatment purposes. On gross examination, the nodule measured 1.4 cm, was well-circumscribed and had a pink-tan coloration. Representative histologic images are shown in figures 1 to 4 (figure 1 at 2X, figure 2 at 4X, and figures 3 and 4 at 10X). Figure 5 is a representative image of the AE1/AE3 immunohistochemical stain.
Q1. Based on the clinical information and histologic findings, what is the most likely genetic alteration?
- ESRW1::ATF1 fusion
- ESWR1::NR4A3 fusion
- EWSR1::CREB1 fusion
- KRAS G12D
Q2. Which of these diagnoses is not part of the differential diagnosis?
- Extraskeletal myxoid chondrosarcoma
- Myoepithelioma
- Poorly differentiated squamous cell carcinoma
- Metastatic melanoma
Q3. The thoracic surgeon asks you what the next appropriate steps for the care of this patient are. What is the most appropriate answer?
- Adjuvant chemotherapy and radioatherapy.
- Imaging and clinical follow-up.
- No folloup necessary, the patient is cured with a resection
- Immunotherapy
Q4. Which of the following immunohistochemical stains is specific to this diagnosis?
- EMA
- Desmin
- S100
- None of the above
Answers to Quiz
Q2. C
Q3. B
Q4. D
Diagnosis
Discussion
Primary pulmonary myxoid sarcoma with EWSR1::CREB1 fusion is a separate entity in the latest WHO Classification of Thoracic Tumors, classified with the mesenchymal tumors specific to the lung. It is a low-grade malignancy affecting mostly middle-aged adults, with a slight predominance towards women, as seen in this case. Many tumors are found incidentally, but a clinical presentation linked with their endobronchial location such as cough or dyspnea is also frequent. A relationship with smoking has not been established.
Grossly, pulmonary myxoid sarcomas measure between 1.5 and 14 cm, are well-delineated and nearly always found in relation to an airway. Under the microscope, they frequently show at least a partial fibrous pseudocapsule with a lymphoplasmacytic component. The tumor is frequently lobulated and is composed of spindle/stellate to round cells arranged in cords within a myxoid stroma. Cellularity is variable and cellular areas within a collagenous stroma can be seen. Mitotic count is usually low. Most tumors are cytology bland, but some cases have been reported with atypia, necrosis, and higher mitotic count. By immunohistochemistry, there is no specific diagnostic marker. These tumors are consistently negative for keratins, S100, neuroendocrine markers, desmin, TTF1, and CD34. Some cases are positive for EMA and most are vimentin positive. Immunohistochemistry is mostly useful to rule out other diagnoses included in the differential (Question 4).
These tumors classically harbor a ESWR1::CREB1 fusion in more than 80% of cases (Question 1). Lung tumors that lack EWSR1::CREB1 fusion are poorly characterized: other fusions such as ESWR1::ATF1, FUS1::ATF1 have been described, as well as SMARCB1 loss. The classic fusion and the morphology of the tumor is shared by tumors affecting other organs of the body diagnosed as myxoid subtype of angiomatoid fibrous histiocytomas (AFH). Their exact relationship with tumors affecting the lung is unclear. AFH are seen mostly this the dermis and subcutis of children and young adults, but they have been described in other organs such as the central nervous system. They have similar histologic findings, including a dense fibrous pseudocapsule, a peritumoral lymphoplasmacytic infiltrate, blood-filed cystic spaces and multinodular aggregates of histiocytoid and spindle tumor cells with a low mitotic rate.
The differential diagnosis is broad. Indeed, a primary lung sarcomatoid carcinoma with heterologous differentiation must be ruled out as well as metastasis from a melanoma or from a known sarcoma in the past medical history of the patient. Based on morphology, the differential includes also extraskeletal myxoid sarcoma, myoepithelioma, chordoma, and chordoid meningioma (Question 2). Metastasis from a myxoid liposarcoma, myxofibrosarcoma and aggressive angiomyxoma can also be considered.
Most reported tumors have an indolent behavior, with no recurrence after resection, but rare cases with progression to a metastatic disease were described. (Question 3). Atypical histologic features (necrosis, higher mitotic count) and absence of the classic ESWR1::CREB1 fusion could correlate with prognosis, although more data is needed. There are no specific recommendations about clinical management due to the rarity of these tumors, but imaging and clinical follow-up after surgical resection could be suggested.
Take home message
Primary pulmonary myxoid sarcoma with EWSR1::CREB1 fusion are rare tumors, with a mostly indolent behavior with a metastatic potential. They typically affect young patients and are nearly always found in association with an airway. Immunohistochemical stains are mostly useful to rule out other entities in the differential. Most of the tumors have a EWSR1::CREB1 fusion which can be diagnosed by FISH.
References
Smith SC, Palanisamy N, Betz BL, et al. At the intersection of primary pulmonary myxoid sarcoma and pulmonary angiomatoid fibrous histiocytoma: observations from three new cases. Histopathology. 2014;65(1):144-6.
Schaefer IM, Fletcher CD. Myxoid variant of so-called angiomatoid "malignant fibrous histiocytoma": clinicopathologic characterization in a series of 21 cases. Am J Surg Pathol. 2014;38(6):816-23.
Thway K, Nicholson AG, Wallace WA, et al. Endobronchial pulmonary angiomatoid fibrous histiocytoma: two cases with EWSR1-CREB1 and EWSR1-ATF1 fusions. Am J Surg Pathol. 2012;36(6):883-8.
Thway K, Nicholson AG, Lawson K, et al. Primary pulmonary myxoid sarcoma with EWSR1-CREB1 fusion: a new tumor entity. Am J Surg Pathol. 2011;35(11):1722-32.
Contributors
Thoracic Pathology and Research fellow
Brigham and Women’s Hospital and Dana-Farber Cancer Institute
Boston (MA), USA
Lynette M. Sholl MD
Thoracic Pathology
Brigham and Women’s Hospital
Boston (MA), USA